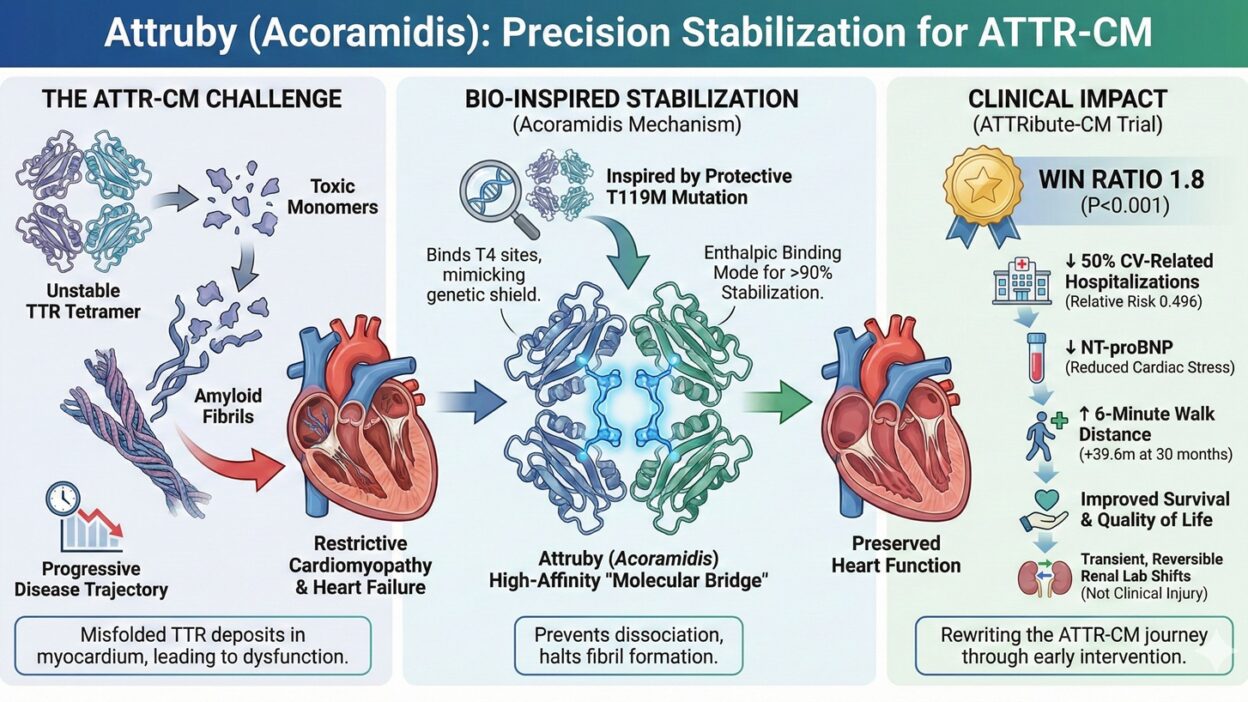
Disclaimer: This blog is for educational purposes only and does not offer medical advice, diagnosis, or treatment. Always consult a healthcare provider for medical concerns.For decades, transthyretin amyloid cardiomyopathy (ATTR-CM) was a daunting diagnostic “dead end” in cardiology. This progressive, life-threatening condition acts as a silent architect of heart failure, where unstable transthyretin (TTR) proteins—normally responsible for transporting thyroxine and retinol—dissociate from their tetrameric form, misfold, and aggregate into toxic amyloid fibrils. These fibrils infiltrate the myocardium, leading to a stiffened, restrictive heart that eventually loses its capacity to support systemic circulation.However, we are witnessing a paradigm shift. With the introduction of Attruby (acoramidis), clinicians now have a high-affinity TTR stabilizer that moves beyond mere symptom management to target the molecular foundation of the disease.
However, we are witnessing a paradigm shift. With the introduction of Attruby (acoramidis), clinicians now have a high-affinity TTR stabilizer that moves beyond mere symptom management to target the molecular foundation of the disease.
The Visionary Behind the Science: The BridgeBio Mission
To understand the breakthrough of Attruby, one must look at its parent company, BridgeBio Pharma. Founded in 2015 with a “patient-first” mandate, BridgeBio was built specifically to bridge the gap between academic discovery and clinical delivery for genetic diseases.
- A “String of Pearls” Approach: Unlike traditional pharmaceutical giants, BridgeBio operates with a unique “hub-and-spoke” model. This allows dedicated, lean teams of experts to focus exclusively on specific genetic drivers—such as TTR—ensuring that every decision is guided by deep domain expertise rather than corporate bureaucracy.
- Driven by Genetic Truth: The company’s philosophy is rooted in finding the most direct path to treating the cause of a disease. This pursuit led them to the T119M protective mutation, which became the structural inspiration for acoramidis.
- A Commitment to Access: Innovation only matters if it reaches the patient. Through programs like ForgingBridges, BridgeBio actively works to remove financial and insurance barriers, providing support for co-pays and insurance navigation to ensure therapy is accessible to those who need it most.
The TTR Stabilizer with a Genetic Pedigree
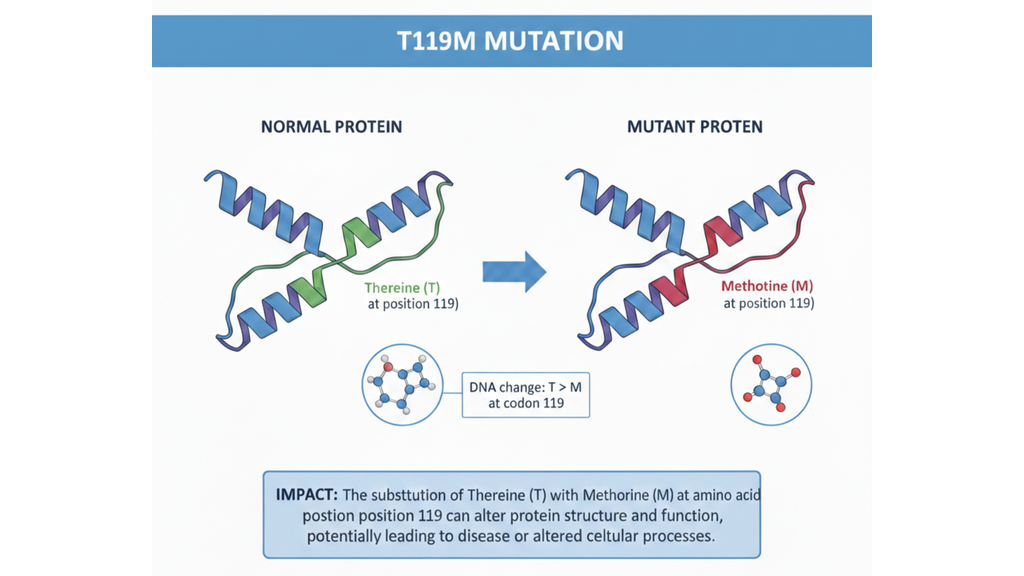
The development of Attruby is more than just chemistry; it is a masterclass in rational drug design inspired by nature itself. Scientists observed a phenomenon in individuals born with a rare mutation known as T119M. This mutation acts as a genetic “shield,” naturally stabilizing the TTR protein through unique hydrogen bonding and protecting carriers from the development of amyloidosis.
Attruby was specifically engineered to mirror this natural advantage:
- High-Affinity Anchoring: The molecule binds directly to the two T4 binding sites on the TTR tetramer, holding the protein together like a molecular bridge to prevent dissociation into monomers.
- The Enthalpic Advantage: While earlier stabilizers relied on entropic (hydrophobic) binding, Attruby utilizes a primarily enthalpic binding mode. This hydrogen-bond-heavy approach significantly enhances both its binding affinity and its potency compared to older therapeutic options.
Total Stabilization: Clinical data demonstrated that Attruby achieves near-complete stabilization (>90%) of both wild-type and variant TTR across the entire dosing interval. This level of stabilization is critical, as greater TTR stabilization is correlated with improved clinical outcomes.
Dissecting the Evidence: The ATTRibute-CM Trial
In the landmark Phase 3 ATTRibute-CM trial, 632 patients were randomized in a 2:1 ratio to receive either 800 mg of acoramidis twice daily or a placebo. To capture the full clinical picture over a 30-month duration, researchers employed a rigorous four-step hierarchical analysis using the Finkelstein-Schoenfeld method.The trial yielded a powerful win ratio of 1.8 ($P < 0.001$), demonstrating that patients on acoramidis were consistently “winning” across mortality, morbidity, and physical function.
| Hierarchical Component | Clinical Significance |
|---|---|
| CV-Related Hospitalization | Achieved a 0.496 relative risk ratio, essentially cutting the frequency of hospital visits in half. |
| NT-proBNP Change | Demonstrated the highest win-to-loss ratio (23.3% vs. 7.0%), serving as a clear signal of reduced cardiac wall stress and heart failure progression. |
| 6-Minute Walk Distance | Patients on Attruby walked an average of 39.6 m further than the placebo group at 30 months, representing a significant preservation of functional capacity. |
| Death from Any Cause | This critical endpoint, along with hospitalizations, contributed more than half of the total “wins” in the study. |
Safety and the “Renal Shift” Reality
For the modern clinician, a drug’s safety profile is the bedrock of long-term adherence. In the trial, Attruby was shown to be well-tolerated, with an overall incidence of adverse events similar to placebo (98.1% vs. 97.6%).
- Gastrointestinal Health: Diarrhoea was noted in 11.6% of patients (compared to 7.6% for placebo), and upper abdominal pain was reported in 5.5% (vs. 1.4% for placebo). Most cases were mild and did not lead to drug discontinuation.
- Renal Lab Observations: A specific laboratory trend was noted: within the first 4 weeks, patients exhibited a mean increase in serum creatinine of 0.2 mg/dL and a mean decrease in estimated glomerular filtration rate (eGFR) of 8.2 mL/min/$1.73~m^2$.
A Noteworthy Distinction: Crucially, these shifts stabilized after Day 28 and were found to be reversible if treatment was discontinued. This suggests the changes represent a pharmacological lab effect (likely related to transporter inhibition) rather than clinical kidney injury.
Clinical Practice: The Path Forward
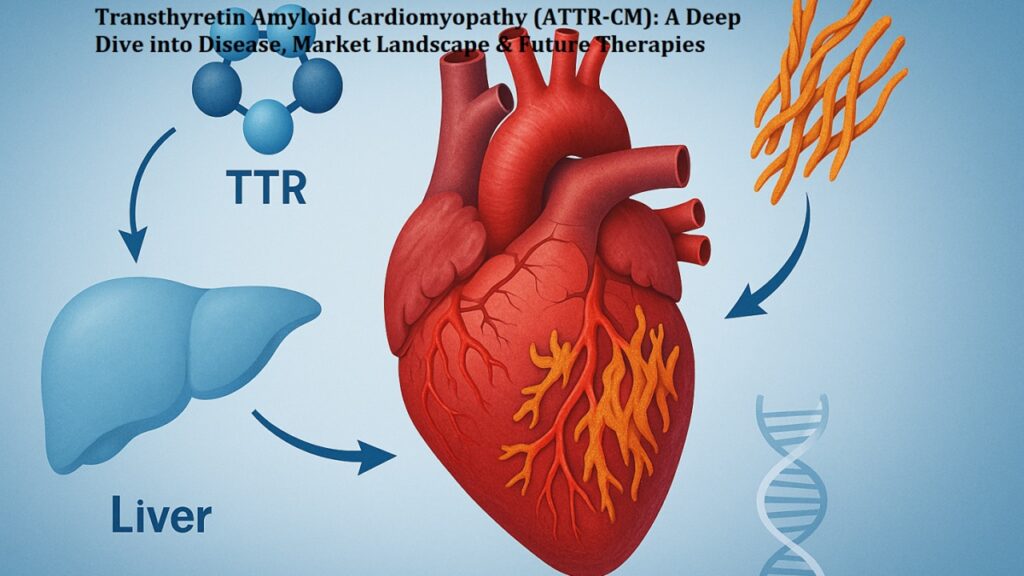
As non-invasive diagnostic algorithms—such as technetium-99m scintigraphy—become the gold standard, we are identifying ATTR-CM patients earlier than ever before. This early detection window is critical because the goal of stabilization is to preserve existing myocardium before irreversible damage occurs.Attruby offers a robust therapeutic intervention that begins stabilizing TTR in as early as 28 days. By preventing the dissociation of TTR into toxic monomers, clinicians can now intervene effectively in both wild-type and variant cases, fundamentally rewriting the trajectory of the disease and improving the quality of life for their patients.
Access the Source: Efficacy and Safety of Acoramidis in Transthyretin Amyloid Cardiomyopathy